
To get on a two degrees path carbon emissions from coal need to be cut by two-thirds of today’s levels by 2040. That isn’t only about closing coal plants…It is also about developing a global [carbon capture and storage] industry that must by mid-century become bigger than the world’s oil extraction industry.
— Carbon Brief, 12/9/14
As we enter 2015, the world has a good tally of the carbon left to burn before crossing the 2°C level agreed upon as safe. Because it will take decades to replace our global energy system, plans to achieve 2°C now rely on getting carbon back out of the air, at some point in the future, and storing it deep underground.
Carbon capture and storage (CCS) would be a vast industry, and would actually need to handle more material than the oil industry currently moves in order to recover what we burn on a daily basis.
The amount of CO2 waste is bigger than the carbon fuel itself for reasons known to high school chemistry: carbon has an atomic weight of 12; oxygen of 16.
When we burn a fossil fuel, one carbon atom combines with two oxygen atoms to form a CO2 molecule that combines these atomic weights, to equal 44. What this means in everyday life is that while a gallon of gasoline, mostly carbon, weighs about six pounds, it turns into over 19 pounds of CO2 after your car engine burns it.
The CO2 waste gas is invisible at the tailpipe, so it dissipates unnoticeably into the air, but changes the heat absorption of the atmosphere via the greenhouse effect.
New technology might capture that emitted CO2 from the atmosphere, and then possibly compress it back to a liquid at the roughly the density of gasoline in order to be safely buried deep underground.
The volume of liquid CO2 to bury measures a bit more than three times larger than the volume of gasoline you pumped into the car in the first place. And all our fossil fuel cars, jets, power plants and buildings would need to be matched by capture systems handling more than triple the material than in the fuels themselves, in order to bring them to carbon neutrality, which we must reach by later this century in order to hold to the 2C target.
Understanding the challenge of geoengineering may be one way to encourage people to radically reduce emissions, because it’s not easy (or cheap) to put the genie back in the bottle. As climate scientist Ken Caldeira has said, “It is time to stop building things with tailpipes and smokestacks.”
William Wepsala reports on a researcher who is at the forefront of carbon capture, Peter Kelemen of the Earth Institute at Columbia University.
What if the key to halting climate change lay not in the atmosphere above your head, but in the rock beneath your feet? This is a question that Dr. Peter Kelemen has been asking himself for the past ten years.
Kelemen is a geologist at Columbia University’s Earth Institute. His interest in climate change and the Earth’s geology cross paths in a relatively common igneous rock known as peridotite.

Kelemen first came to study peridotite in Oman in the 90s. The element is typically buried in the Earth’s mantle, though tectonic processes pushed large pieces up to the surface in certain areas, including Oman. Studying it, he believed, would give insight into the geology of the Earth’s mantle.
But he discovered another interesting characteristic of peridotite instead. The rock reacts rapidly with carbon dioxide in the atmosphere, trapping it in a solid substance known as carbonate. This carbonation was at first confounding, obscuring the peridotite with white carbonate deposits, making study difficult. Eventually, annoyance gave way to curiosity as Kelemen pondered the potential for carbon storage as a means to slow global climate change.
Geoengineering as a concept is beginning to gain traction among climate scientists. It is unlikely that emissions cuts will equal what the International Panel on Climate Change says is necessary to keep global warming to two degrees celsius. That does not mean that the two degree goal is unattainable.
[pullquote align=”right”]The plan: drill into peridotite formations, pump CO2-rich sea water into the holes, and carbonation will form limestone as the water reacts with the rock, making stable storage for the carbon. [/pullquote]Harnessing geological processes is not an idea unique to Kelemen. The New York Times recently published an article profiling Dutch geochemist, Dr. Olaf Schuiling, and his work with olivine, a reactive mineral within peridotite. Schuiling hopes to speed up the rock CO2 weathering process by crushing the rock and spreading it in areas such as beaches, pathways, and dikes.
Kelemen believes peridotite has the capability to store more CO2 at a faster rate by drilling into natural layers of rock in the Earth’s surface. In Oman, these bore holes will be injected with sea water saturated with CO2; with this method, the gas will be more efficiently exposed to the surface of the rock, and more CO2 will be removed.
A former colleague of Kelemen’s at the Earth Institute, Dr. Klaus Lackner, works on a complementary device to rapidly capture carbon from the air and pipe it underground, where it could be sequestered within carbonated peridotite. (Dr. Lackner is interviewed here in City Atlas). Last year, Lackner left Columbia to take up a post at Arizona State University, enticed, he said, by the opportunity to “build stuff in the desert” – the dry desert environment being an ideal surrounding for Dr. Lackner’s experimental air capture devices.
In the fall, Lackner came back to Columbia to participate in a panel discussion on the climate-energy nexus. Accompanied by other energy experts, Lackner stressed the importance of carbon capture and storage. He maintained that renewable energy in the form of solar and wind is promising, but ultimately unable to meet demand in lieu of fossil fuels, especially when taking into consideration the growing demand from countries like India and China. Because fossil fuel will be needed to meet global energy needs, greenhouse gas emissions will likely not be cut enough to achieve the 2 degrees celsius goal.
A way to integrate Lackner’s points on carbon capture and Kelemen’s work on peridotite is to implement on-site capture and storage. Constructing on or near peridotite deposits could accomplish this. Capturing CO2 at the point of emission (for instance, a coal power plant) would stop the gas from being emitted into the atmosphere. Storing it in water and injecting it into bore holes drilled in the peridotite could be a viable means of disposal.
A big benefit of Kelemen’s geologic method of storing carbon as a stable solid is that it removes the risk of a bubble of pressurized CO2 leaking from an underground storage site.
So if it is achievable and the technology exists, why isn’t it being implemented? Like many other policy issues, this can be explained in a word: money.
Currently, the technology to capture CO2 at the source of emission (e.g. a coal-burning power plant) is in the laboratory. At its current iteration, it is too expensive to implement without any incentive to do so. As the technology improves, though, economies of scale will be achieved and costs will drop. The issue of incentive could be remedied through policy. Governments could simply mandate that emissions be limited, or carbon stored. A marketplace could also be set up to trade emissions credits.
Kelemen’s plan also suffers from a dearth of readily available funds. At the beginning of his studies in Oman, Shell put money into the project as a part of its sustainability agenda. The company has since pulled its money out of the project. Kelemen says he needs $10 million to conduct a feasibility study of the project.
The Omani government has been a willing participant in the project as it sees it as a potential source of revenue if an emissions trading scheme is developed, or if they are paid to store CO2 captured elsewhere. That said, the government is wary of turning into the world’s dumping ground and all the baggage that accompanies it.
Kelemen is indomitable. Despite lacking the funds to do a complete study, Kelemen and his team are laying the foundation for potential future study. The Oman Drilling Project will be carried out next winter. Instead of completing all phases, the team will be drilling the bore holes needed for a complete study to specification. The idea is that, if additional funding materializes, the project will already be in place for phase two of the operation, injecting CO2-laden water to store carbon.
Funding may not be far off. The New York Times piece on Dr. Schuiling reported that the National Academy of Sciences will publish a special report on geoengineering early this year. This may well be the beginning of a paradigm shift in the debate on climate change, with money and support for those with plans to apply human ingenuity to the problem. As the IPCC and other institutions publish increasingly ominous predictions, scientists like Kelemen are rolling up their sleeves.
View a photo essay of Dr. Kelemen’s work in Oman.
For a related look at the upside and downside of geoengineering, see British physicist David MacKay’s excellent guide to energy.
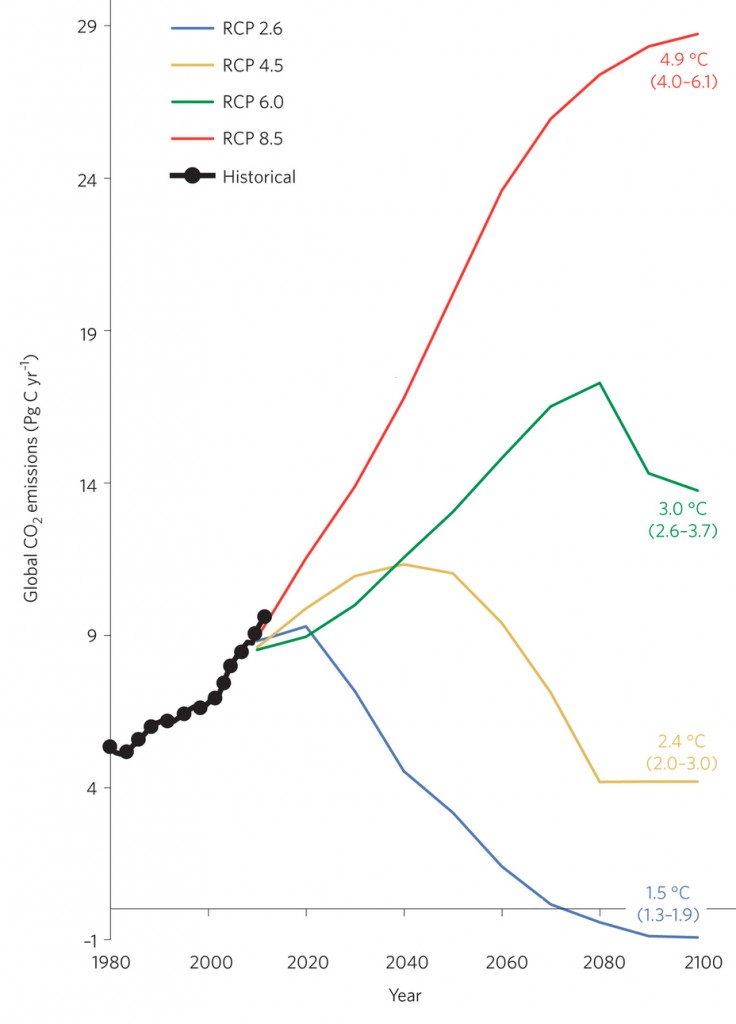
As a reminder of urgency, the Bulletin of the Atomic Scientists has moved their ‘doomsday clock’ forward to three minutes to midnight. The Bulletin also features an earlier, detailed overview of the challenges of climate change. As in the chart at the top of this page, the longer we wait to reduce emissions, the faster the drop has to be.
